論文が、Organic Electronicsに掲載されました。ぜひ、ご覧ください。
Side-chain engineering of oligothiophene-extended triphenylamines for stable radical cations with NIR-II to NIR-III absorption
Masafumi Yano , Koji Yamashita , Minori Yano , Yoshitsugu Komai , Yuki
Arikata , Koichi Mitsudo , Yukiyasu Kashiwagi
https://doi.org/10.1016/j.orgel.2026.107415
Abstract
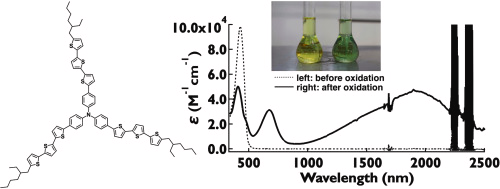
Bithiophene- and terthiophene-appended triphenylamine (TPA) derivatives were synthesized and systematically investigated. Upon one-electron oxidation, these compounds generated stable radical cations exhibiting intense near-infrared (NIR) absorption. The bithiophene derivatives combined sufficient solubility with strong NIR-II absorption, whereas the corresponding terthiophene analogues showed further bathochromic shifts but suffered from extremely poor solubility. Incorporation of branched alkyl substituents effectively restored solubility. Notably, the branched terthiophene radical cation displayed an absorption maximum beyond 1900 nm, representing one of the longest wavelengths reported for TPA-based radical cations. These findings establish π-extension and side-chain engineering as complementary strategies for the rational design of soluble and stable radical-cation dyes with NIR-II to NIR-III absorption.